Interested in volunteering for research? Fill out this survey to be considered for current and future studies: Go to Survey
Or contact us at (850) 644-2824 / FSUN@med.fsu.edu
Actively Recruiting
Virtual Reality Mindfulness and Accelerated TMS Treatment for Individuals with PTSD
Purpose: Treatment study aiming to use technology to improve the treatment of PTSD.
- Age Range: 18-65
- Condition: Individuals with PTSD
- Treatment Protocol:
- Virtual Reality Guided Mindfulness: once per day for 10 days (two weeks).
- (If symptoms persist) Accelerated transcranial magnetic stimulation (TMS) treatment Week 1: 5 sessions per day for 5 days. Participants are randomized into active or sham TMS.
- Accelerated TMS Week 2: 5 sessions per day for 5 days. All participants will receive active TMS treatment during this week.
- Three follow-up visits at 1-, 3-, and 6-months post treatment.
- Some compensation provided for evaluation visits.
Personalized TMS Dosing for Individuals with PTSD (NON-TREATMENT)
Purpose: Technology development study aiming to improve the treatment of PTSD.
- Age Range: 18-70
- Condition: Individuals with PTSD
- Protocol:
- This study uses Transcranial Magnetic Stimulation (TMS) and a measure of brain activity called functional Near-Infrared Spectroscopy (fNIRS).
- Three study visits:
- Initial office visit for evaluation and TMS motor threshold
- MRI scan of your brain
- Simultaneous TMS and fNIRS testing session
- This is NOT a treatment study, so compensation will be provided for your time.
Quality Improvement Survey: Community Perception of Neuromodulation Research
We created a quality improvement survey available to all members of the greater Tallahassee community to give us feedback on a variety of aspects of the type of research that we do. Our primary goal is to identify and engage community partners, particularly those representing groups with unmet mental health needs. We would greatly appreciate if you took a few minutes of your time to fill out our survey! Available at this link: click here . You may also scan the QR code with your device. All responses are anonymous and no identifying information will be gathered.

MagVenture MagPro R30 TMS Machine
More info can be found at MagVenture's website .
Magstim Rapid 2 TMS Machine

More info can be found at MagStim's website .
Brainsight® Neuronavigation with Specialized TMS Chair
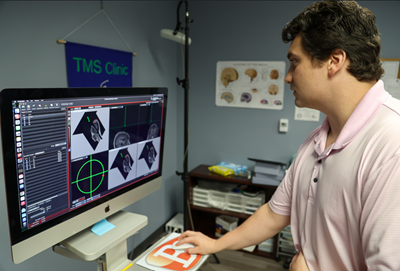
More info can be found at Brainsight's website .
NIRx NIRScout fNIRS System
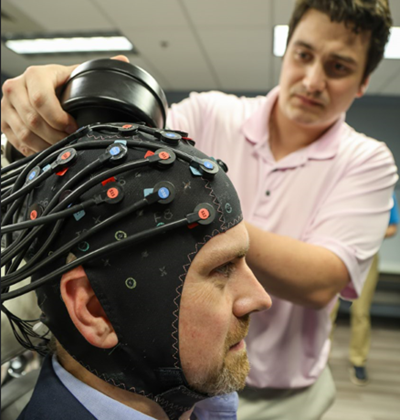
More info can be found at NIRx's website .
Valve Index VR System and Oculus Quest 2 VR System

More info can be found at the Valve and Oculus websites.
Computers/Software
-
Lenovo ThinkPad laptops and monitors
-
27” iMac for the Brainsight Neuronavigation system
-
Office software: Microsoft Office, Adobe
-
Statistical/Research software: MATLAB, SPSS, SAS, PsychoPy, NIRx
Neuromodulation for Depression, Anxiety, PTSD and Pain
Purpose: Treatment study aiming to improve symptoms related to Anxiety, Depression, PTSD and Chronic Pain.
- Age range: 18+
- Condition: Individuals with Depression, Anxiety, PTSD, and/or Chronic Pain
- Treatment Protocol:
- Virtual Reality Guided Mindfulness: once per day for 10 days (two weeks). Five weekly follow-up sessions if you experience a benefit from treatment.
- (If symptoms persist) Accelerated transcranial magnetic stimulation (TMS) treatment Week 1: 5 sessions per day for 5 days. Five weekly follow-up sessions if you experience a benefit from treatment.
- (If symptoms persist) Accelerated TMS Week 2: 5 sessions per day for 5 days. Five weekly follow-up sessions.
Neuromodulation for Postpartum Depression
Purpose: Treatment study aiming to improve symptoms related to Postpartum Depression.
- Age range: 18+
- Condition: Individuals with Postpartum Depression
- Treatment Protocol:
- Virtual Reality Guided Mindfulness: once per day for 10 days (two weeks). Five weekly follow-up sessions if you experience a benefit from treatment.
- (If symptoms persist) Accelerated transcranial magnetic stimulation (TMS) treatment Week 1: 5 sessions per day for 5 days. Five weekly follow-up sessions if you experience a benefit from treatment.
- (If symptoms persist) Accelerated TMS Week 2: 5 sessions per day for 5 days. Five weekly follow-up sessions.
Innovative Treatment Trial for Veterans with PTSD
Purpose: Treatment study aiming to help us learn more about Post-Traumatic Stress Disorder (PTSD) and test whether repetitive Transcranial Magnetic Stimulation (rTMS) alone, Cognitive Processing Therapy (CPT) alone, or rTMS combined with CPT can improve clinical outcomes.
- Age range: 18-60
- Condition: Veterans with PTSD
- Treatment Protocol:
- Baseline visits: Three in-office visits for rating scales, an EEG, and determining dose for treatment.
- Treatment: Participants are randomized to a treatment condition, and treatment is once/week for 12 weeks.
- Follow Up: Three follow-up visits over 6 months post-treatment.
Characterizing Scalp Tolerability of TMS (NON-TREATMENT)
Purpose: To get a better understanding of the difference in how Transcranial Magnetic Stimulation (TMS) feels on different parts of the scalp. This is not a treatment study.
- Age range: 18+
- Condition: N/A
- Study Protocol:
- There is one study visit in which participants will undergo a brief safety evaluation and complete rating scales, and be stimulated on various parts of the scalp with the TMS coil. Compensation will be provided for your time.